These industries use QM Pilot
Health Care System
Hotels/Eventmanagement
Education
Public Administration
Industry
Service Sector
Associations & organizations
Other sectors
"We no longer waste time searching for documents, and there are no old versions floating around! Having all the latest information stored in one place in a structured way makes our everyday life much easier - exactly what we need."
Uschi Miller, Therwil
Other clients in Health Care Systems:
Other clients in Hotels/Eventmanagement:
Other clients in Education:
"The well thought-out and clean internal logic of QM Pilot enabled our members of the local council, who work in the militia system, to familiarise themselves quickly."
Dr. Michael Gisler, Gemeinderat, Finanzvorsteher Mauensee
Other clients in Public Administration:
Other clients in Industry:
"Thanks to QM Pilot, we can record and manage processes and applicable documents much more efficiently. The tool meets our requirements 100% and we are thrilled with it!"
Andreas Helfenstein, Zürich
Other clients in Service sector:
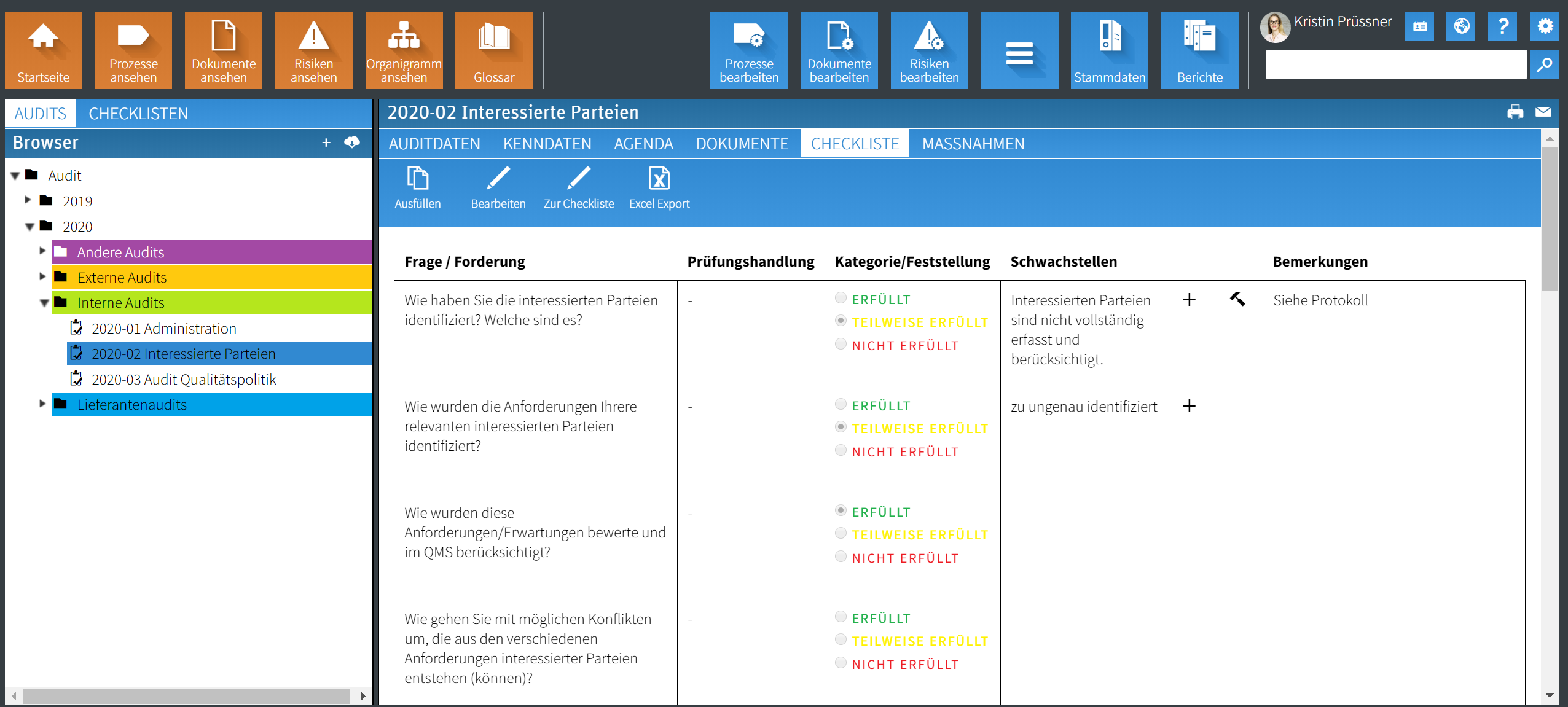
"During the introduction of the QMS, a better understanding and broad acceptance of quality management was created among management and employees.
Swisstransplant, Use Case
Other clients in Associations & organizations:
"With QM Pilot, we have obtained a lean and user-friendly solution. The intuitive presentation as well as the lean management, guidance and control of processes and documents increase information transparency and simplify administrative processes."
Swiss Prime Site, Use Case
Other clients in Other sectors:






.jpg)


























 ist ein Produkt der
ist ein Produkt der 